Announcing the New Mock OB/GYN Shelf/Clerkship Exam

Introducing the Mock OB/GYN Shelf/Clerkship Exam, best suited for medical students who want to excel in their OB/GYN clerkship.
The Mock OB/GYN Shelf/Clerkship Exam is located in your Boost Box (on the home page of your desktop/laptop Rosh Review account). It contains 100 questions and comprehensive explanations to help you prepare for the NBME Obstetrics & Gynecology Shelf Exam.
Categories covered include topics such as hypertensive disorders, gynecologic cancers, and contraception with a few “zebras” to keep you on your toes.
Here is an example of a question and explanation:
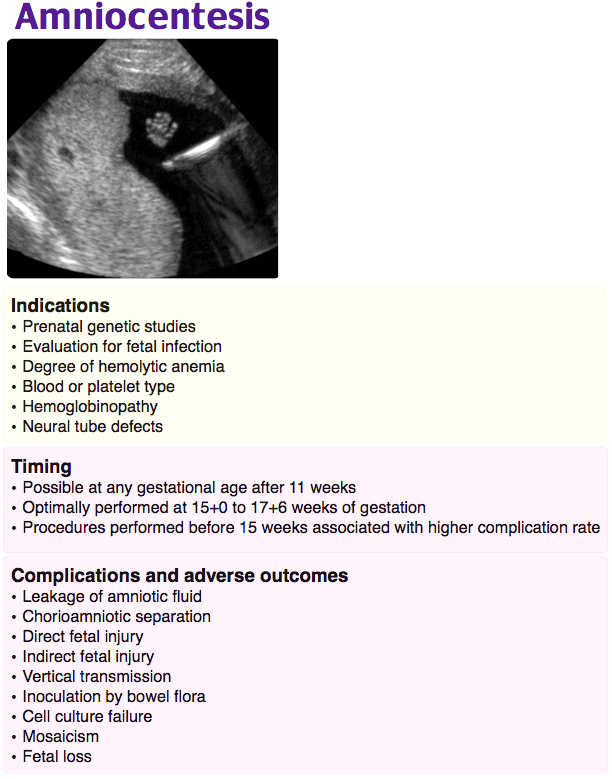
A 38-year-old woman, G3P2, at 15 weeks gestation presents to the office for routine prenatal care. Her first two pregnancies resulted in deliveries of two healthy children. Cell-free fetal DNA test results show an increased risk for trisomy 13. The patient asks what the next step in evaluation would be. Which of the following tests is the most appropriate to recommend to this patient?
A. Amniocentesis
B. Chorionic villus sampling
C. Maternal serum quadruple test
D. Nuchal translucency ultrasound
Answer A
During pregnancy, the apoptosis of syncytiotrophoblastic placental cells leads to the presence of extracellular fetal DNA in the maternal circulation. The cell-free fetal DNA test (also known as noninvasive prenatal testing) screens for fetal aneuploidy (e.g., trisomy 13, 18, 21) by sequencing this fetal DNA. It can also be used to determine the fetal sex and Rh status. The test performance is dependent on the concentration of cell-free fetal DNA, the fetal fraction, in the maternal blood. Testing can be performed starting at 10 weeks gestation when the fetal fraction is sufficient to avoid testing failure. Although cell-free fetal DNA testing has a high detection rate for autosomal and sex chromosomal aneuploidy, it is considered a screening test due to performance limitations including a small but significant risk of false positives (due to maternal or placental mosaicism or a demised twin) and false negatives. Cell-free fetal DNA is currently recommended for patients at high risk for aneuploidy (e.g., maternal age > 35, abnormal biochemical screening test for trisomy, abnormal fetal anatomy ultrasound). Positive screening test results are an indication for invasive diagnostic testing. In this patient, the most appropriate diagnostic test is amniocentesis, which involves the ultrasound-guided needle aspiration of amniotic fluid to sequence and karyotype the fetal DNA directly from amniocytes, fetal cells floating in the amniotic fluid.
Chorionic villus sampling (B) is an invasive prenatal diagnostic genetic test that can be offered to patients who have positive prenatal genetic screening tests. However, it is typically performed from 10 to 13 weeks gestation. The maternal serum quadruple test (C) is a screening test for trisomy 18, trisomy 21, and neural tube defects. It is not used as a diagnostic follow-up to other prenatal screening tests. Ultrasound measurement of nuchal translucency (D) is a component of the first trimester combined test, a test that screens for trisomy 18 and 21 and is performed from 9 to 13 weeks gestation.
One Step Further question:
The first-trimester screen uses which maternal serum markers?
Answer:
Beta-hCG and pregnancy-associated plasma protein-A (PAPP-A).
We also have mock shelf exams available for emergency medicine, internal medicine, family medicine, pediatrics, and psychiatry. And be sure to check out our OB/GYN Qbank for thousands of comprehensive questions.
If you’re interested in How to Increase Your CREOG Exam or ABOG Qualifying Exam Score and other tips, head over to the Rosh Blog.
Study on,
The Rosh Review Team





Comments (0)